The Science Behind UV-C Energy: What Is It? - UV Resources
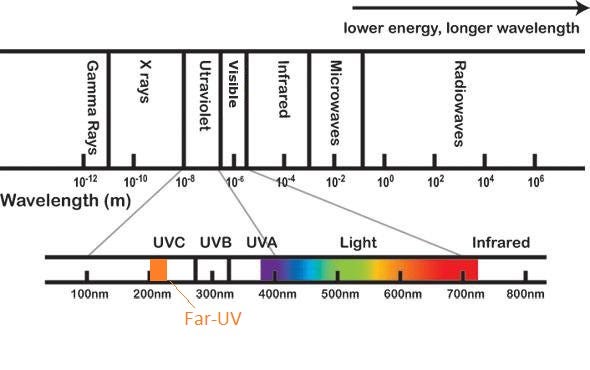
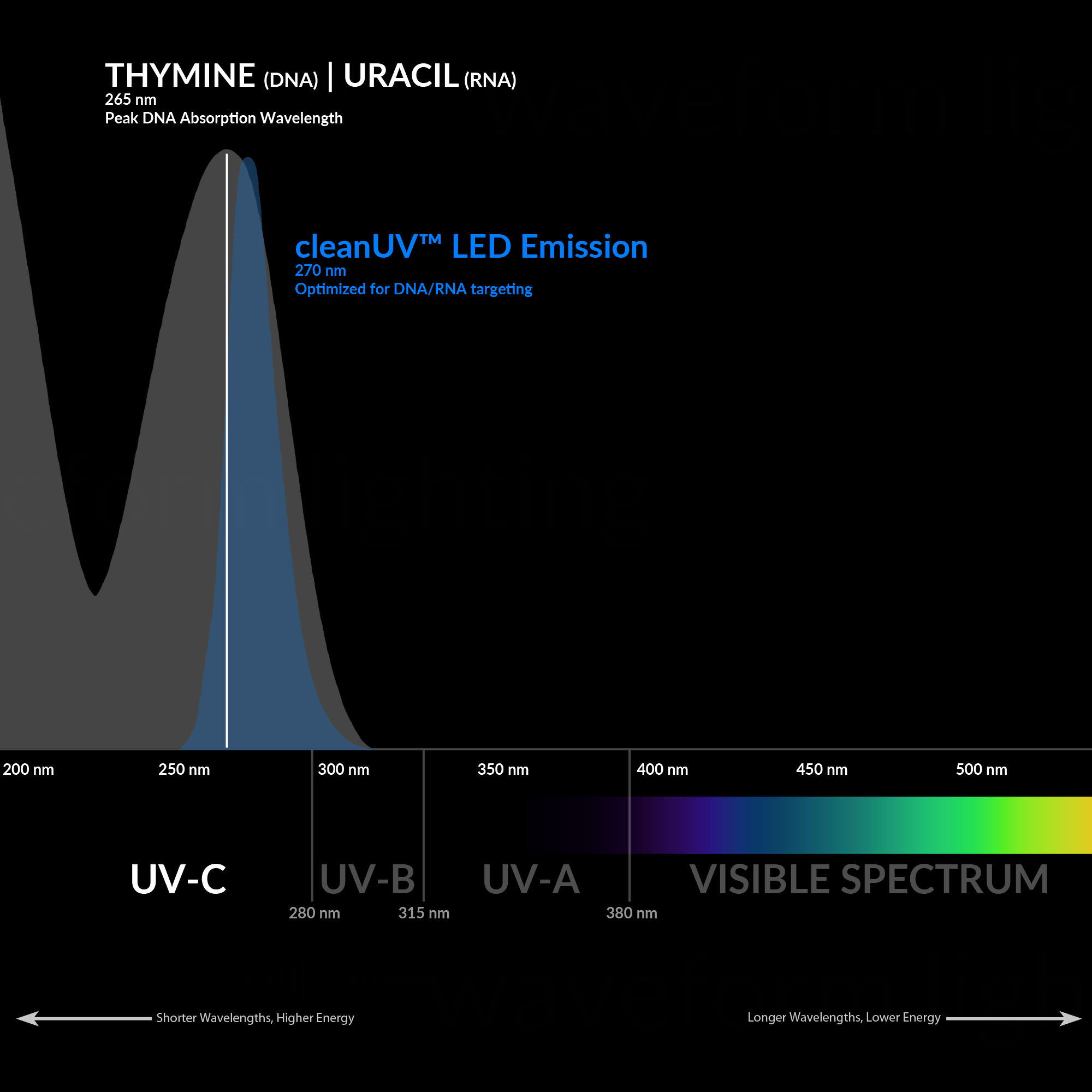
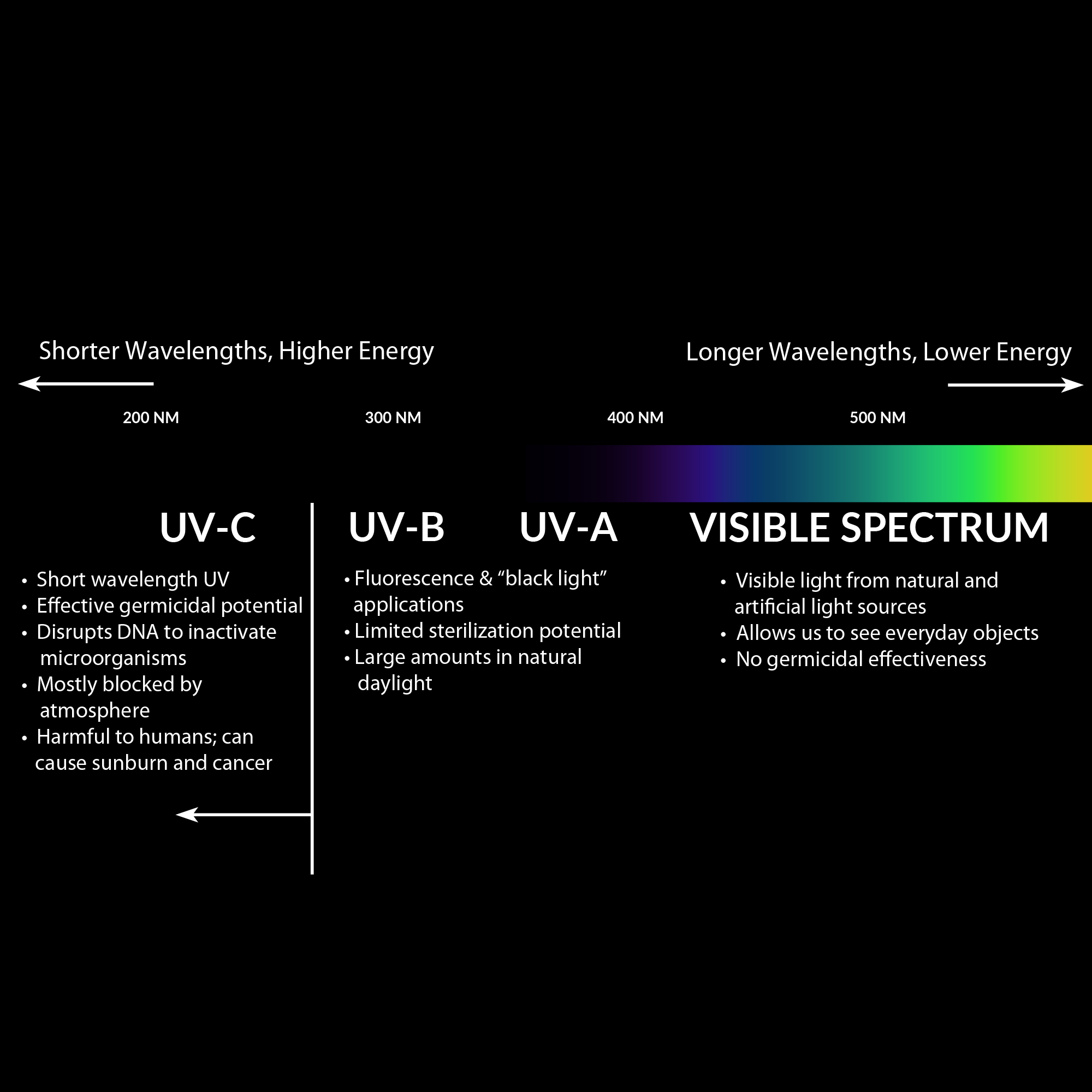
UV light comprises a segment of the electromagnetic spectrum between 400 and 100 nm, corresponding to photon energies from 3 to 124 eV. The UV segment has four sections, labeled UV-A (400 to 315 nm), UV-B (315 to 280 nm), very high energy and destructive UV-C (280 to 200 nm), and vacuum UV. Most of us are familiar with the harmful effects of UV energy transmitted by sunlight in the UV-A and UV-B wavelengths, giving rise to UV “sunburn” inhibitors, or blocking agents, which are found in glasses and lotions. We are also familiar with products engineered to withstand the effects of UV radiation, such as plastics, paints, and rubbers. However, unlike the UV-A and UV-B wavelengths, the UV-C band has more than twice the electron volt energy (eV) as UV-A, and it is well absorbed (not reflected) by organic substances, adding to its destructiveness.

Understanding Ultraviolet Light (UV-C) and Its Benefits - Therma
What is the Difference Between UV-A and UV-C?

The Science Behind UV-C Energy: What Is It? - UV Resources
What is the Difference Between UV-A and UV-C?

Ultraviolet germicidal irradiation - Wikipedia

UVC radiation for food safety: An emerging technology for the microbial disinfection of food products - ScienceDirect

Tempted to buy a UV light disinfection gadget? Some can be dangerous – here's what you need to know
Ultraviolet germicidal irradiation - Wikipedia

How Does UVC Technology Work?

What Is Germicidal Ultraviolet-C (UVC) light?